A rigid tank contains $35 \,\,kg$ of nitrogen at $6$ atm. Sufficient quantity of oxygen is supplied to increase the pressure to $9$ atm, while the temperature remains constant. Amount of oxygen supplied to the tank is .... $kg$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA closed compartment containing gas is moving with some acceleration in horizontal direction. Neglect effect of gravity. Then the pressure in the compartment is
- 2The total internal energy of two mole monoatomic ideal gas at temperature $T=300\,K$ will be ...........$J$.View Solution
$\text { (Given } R =8.31\,J / mol.K )$
- 3If a given mass of gas occupies a volume of $10\, cc$ at $1$ atmospheric pressure and temperature of $100°C(373.15\, K).$ What will be its volume at 4 atmospheric pressure; the temperature being the same .... $cc$View Solution
- 4For one gram mol of a gas, the value of $R$ in the equation $PV = RT$ is nearly ...... $cal/K$View Solution
- 5The molecules of a given mass of a gas have $r.m.s.$ velocity of $200 \,m s^{-1}$ at $27^o C$ and $1.0 \times 10^5 \,Nm^{-2}$ pressure. When the temperature and pressure of the gas are respectively, $127^o C$ and $0.05 \times 10^5 \,Nm^{-2},$ the $r.m.s.$ velocity of its molecules in $m s^{-1}$ isView Solution
- 6A system consists of two types of gas molecules $A$ and $B$ having same number density $2 \times$ $10^{25}\, / {m}^{3}$. The diameter of ${A}$ and ${B}$ are $10\, \stackrel{\circ}{{A}}$ and $5\, \stackrel{\circ}{{A}}$ respectively. They suffer collision at room temperature. The ratio of average distance covererd by the molecule $A$ to that of $B$ between two successive collision is $.....\,\times 10^{-2}$View Solution
- 7A vessel is partitioned in two equal halves by a fixed diathermic separator. Two different ideal gases are filled in left $(L)$ and right $(R)$ halves. The rms speed of the molecules in $L$ part is equal to the mean speed of molecules in the $R$ part. Then the ratio of the mass of a molecule in $L$ part to that of a molecule in $R$ part isView Solution

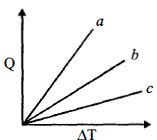
- 8Figure shows the variation in temperature $\left( {\Delta T} \right)$ with the amount of heat supplied $(Q)$ in an isobaric process corresponding to a monoatomic $(M)$, diatomic $(D)$ and a polyatomic $(P)$ gas. The initial state of all the gases are the same and the scales for the two axes coincide. Ignoring vibrational degrees of freedom, the lines $a, b$ and $c$ respectively correspond toView Solution
- 9View SolutionThe molecules of air in the room that you are sitting are all experiencing the force of gravity tending to bring them down. The molecules are also frequently and randomly undergoing collisions, which tend to oppose the effect of fall under gravity. The density of air is nearly uniform throughout the room because
- 10When water is heated from $0$ to $4\,^oC$View Solution
