What is the value of $\frac{R}{{{C_P}}}$ for diatomic gas
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThat gas cannot be liquified
- 2The average kinetic energy of hydrogen molecules at $300 K$ is $E.$ At the same temperature, the average kinetic energy of oxygen molecules will beView Solution
- 3From the following statements, concerning ideal gas at any given temperature $T,$ select the correct one(s)View Solution
- 4$N$ molecules each of mass $m$ of gas $A$ and $2N$ molecules each of mass $2m$ of gas $B$ are contained in the same vessel at temperature $T.$ The mean square of the velocity of molecules of gas $B$ is ${v^2}$ and the mean square of $x$ component of the velocity of molecules of gas $A$ is ${w^2}$. The ratio $\frac{{{w^2}}}{{{v^2}}}$ isView Solution
- 5Which statements are correct about degrees of freedom?View Solution
$A.$ $A$ molecule with $n$ degrees of freedom has $n^{2}$ different ways of storing energy.
$B.$ Each degree of freedom is associated with $\frac{1}{2} RT$ average energy per mole.
$C.$ $A$ monoatomic gas molecule has $1$ rotational degree of freedom where as diatomic molecule has $2$ rotational degrees of freedom
$D$ $CH _{4}$ has a total to $6$ degrees of freedom Choose the correct answer from the option given below:
- 6Five particles have speeds $1, 2, 3, 4, 5 \,\,m/s$. the average velocity of the particles is (in $m/s$)View Solution
- 7A cubical box with porous walls containing an equal number of ${O_2}$ and $H_2$ molecules is placed in a large evacuated chamber. The entire system is maintained at constant temperature $T.$ The ratio of ${v_{rms}}$ of ${O_2}$ molecules to that of the ${v_{rms}}$ of $H_2$ molecules, found in the chamber outside the box after a short interval isView Solution
- 8View SolutionIn kinetic theory of gases, which of the following statements regarding elastic collisions of the molecules is wrong
- 9One litre of Helium gas at a pressure $76\, cm$ of $Hg$ and temperature ${27}^\circ$ is heated till its pressure and volume are doubled. The final temperature attained by the gas is ..... $^oC$View Solution
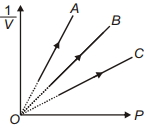
- 10Figure shows the isotherms of a fixed mass of an ideal gas at three temperatures $T_A, T_B$ and $T_C$, thenView Solution