The ratio of work done by an ideal monoatomic gas to the heat supplied to it in an isobaric process is
JEE MAIN 2016, Medium
Efficiency of heat engine is given by
$\eta = \frac{W}{Q} = 1 - \frac{{{C_V}}}{{{C_P}}} = \frac{R}{{{C_P}}} = \frac{R}{{\frac{{5R}}{2}}} = \frac{2}{5}$
$\left( {{C_P} - {C_V} = R} \right)$
For monoatomic gas ${C_P} = \frac{5}{2}R.$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A diatomic gas, having $C_{p}=\frac{7}{2} R$ and $C _{ v }=\frac{5}{2} R ,$ is heated at constant pressure. The ratio $dU : dQ : dW :$View Solution
- 2The coefficient of performance of a Carnot refrigerator working between ${30^o}C$ and ${0^o}C$ isView Solution
- 3An ideal gas heat engine operates in a Carnot cycle between $227^o C$ and $127^o C$. It absorbs $6\,kcal$ at the higher temperature. The amount of heat (in $kcal$) converted into work is equal toView Solution
- 4Two gases of equal mass are in thermal equilibrium. If ${P_a},\,{P_b}$ and ${V_a}$ and ${V_b}$ are their respective pressures and volumes, then which relation is trueView Solution
- 5View SolutionConsider a process shown in the figure. During this process the work done by the system
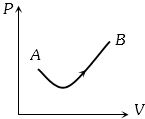
- 6A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?View Solution
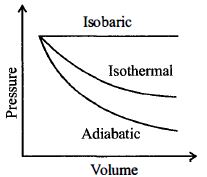
- 7If $\Delta Q$ and $\Delta W$ represent the heat supplied to the system and the work done on the system respectively, then the first law of thermodynamics can be written asView Solution
- 8Find $V_B = ?$View Solution
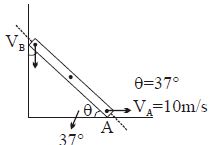
- 9A vessel containing $5\, litres$ of a gas at $0.8 \,pa$ pressure is connected to an evacuated vessel of volume $3$ litres. The resultant pressure inside will be ...... $pa$ (assuming whole system to be isolated)View Solution
- 10View SolutionA given system undergoes a change in which the work done by the system equals the decrease in its internal energy. The system must have undergone an
