The specific heat of $1$ mole of an ideal gas at constant pressure $({C_P})$ and at constant volume $({C_V})$ which is correct
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Open container contains $x$ mole of gas at $T$ $kelvin$ If temperature is tripled, then the amount of gas exit the container isView Solution
- 2Speed of sound in a gas is $v$ and $r.m.s.$ velocity of the gas molecules is $c.$ The ratio of $v$ to $c$ isView Solution
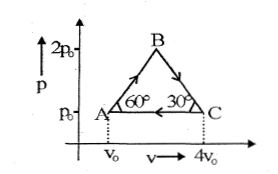
- 3Four moles of an ideal monoatomic gas undergoes a cyclic process $ABCA$ as shown in figure find ratio of temperature of $A$ and $B$ .View Solution
- 4A mixture of ideal gases has $2$ moles of He, $4$ moles of oxygen and $1$ mole of ozone at absolute temperature $T$. The internal energy of mixture is ............. $RT$View Solution
- 5A gas at absolute temperature $300\,K$ has pressure $= 4 \times 10^{-10}\,N /m^2$ . Boltzmann constant, $k = 1.38 \times 10^{-23}\,J / K$ . The number of molecules per $cm^3$ is of the order ofView Solution
- 6One mole of an ideal monatomic gas undergoes a process described by the equation $PV^3 =$ constant. The heat capacity of the gas during this process isView Solution
- 7The temperature at which the $r.m.s.$ speed of hydrogen molecules is equal to escape velocity on earth surface, will be ...... $K$View Solution
- 8Assertion $(A):$ The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5$ times the product of its pressure and volume.View Solution
Reason $(R):$ The molecules of gas collide with each other and the velocities of the molecules change due to the collision.
- 9View SolutionIf the pressure in a closed vessel is reduced by drawing out some gas, the mean free path of the molecules
- 10$Assertion :$ The ratio of $\frac{C_p}{C_v}$ for an ideal diatomic gas is less than that for an ideal monoatomic gas (where $C_p$ and $C_v$ have usual meaning).View Solution
$Reason :$ The atoms of a monoatomic gas have less degrees of freedom as compared to molecules of the diatomic gas.
