Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ The equivalent molar mass of the mixture is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The average thermal energy for a mono$-$atomic gas is : $\left( k _{ B }\right.$ is Boltzmann constant and $T ,$ absolute $e$. temperature)View Solution
- 2If $C_p$ and $C_v$ denote the specific heats of nitrogen per unit mass at constant pressure and constant volume respectively, thenView Solution
- 3If the r.m.s. speed of chlorine molecule is $490\,m / s$ at $27^{\circ}\,C$, the r.m.s. speed of argon molecules at the same temperature will be $......\,m/s$ (Atomic mass of argon $=39.9\,u$, molecular mass of chlorine $=70.9\,u )$View Solution
- 4Same gas is filled in two vessels of the same volume at the same temperature. If the ratio of the number of molecules is $1: 4$, thenView Solution
$A.$ The $r.m.s$. velocity of gas molecules in two vessels will be the same.
$B.$ The ratio of pressure in these vessels will be $1: 4$
$C.$ The ratio of pressure will be $1: 1$
$D.$ The $r.m.s.$ velocity of gas molecules in two vessels will be in the ratio of $1: 4$
- 5Two vessels separately contain two ideal gases $A$ and $B$ at the same temperature, the pressure of $A$ being twice that of $B.$ Under such conditions, the density of $A$ is found to be $1.5$ times the density of $B.$ The ratio of molecular weight of $A$ and $B$ isView Solution
- 6The figure shows the volume $V$ versus temperature $T$ graphs for a certain mass of a perfect gas at two constant pressures of ${P_1}$ and ${P_2}$. What interference can you draw from the graphsView Solution
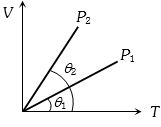
- 7View SolutionAccording to kinetic theory of gases,
- 8Two identical bodies are made of a material for which the heat capacity increases with temperature. One of these is held at a temperature of $100^{\circ} C$, while the other one is kept at $0^{\circ} C$. If the two are brought into contact, then assuming no heat loss to the environment, the final temperature that they will reach isView Solution
- 9At constant temperature on increasing the pressure of a gas by $5\%$ will decrease its volume by ..... $\%$View Solution
- 10A cylinder made of perfectly non conducting material closed at both ends is divided into two equal parts by a heat proof piston. Both parts of the cylinder contain the same masses of a gas at a temperature $t_0 = 27^o$ and pressure $P_0 = 1$ atm. Now if the gas in one of the parts is slowly heated to $t = 57^oC$ while the temperature of first part is maintained at $t_0$ the distance moved by the piston from the middle of the cylinder will be.... $cm$ (length of the cylinder $= 84\,\, cm$)View Solution
