Two ideal Carnot engines operate in cascade (all heat given up by one engine is used by the other engine to produce work) between temperatures, $\mathrm{T}_{1}$ and $\mathrm{T}_{2} .$ The temperature of the hot reservoir of the first engine is $\mathrm{T}_{1}$ and the temperature of the cold reservoir of the second engine is $\mathrm{T}_{2} . T$ is temperature of the sink of first engine which is also the source for the second engine. How is $T$ related to $\mathrm{T}_{1}$ and $\mathrm{T}_{2}$, if both the engines perform equal amount of work?
JEE MAIN 2020, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$N _{2}$ gas is heated from $300\, K$ temperature to $600\, K$ through an isobaric process. Then find the change in entropy of the gas. $( n =1 mole )$ (in $J/K$)View Solution
- 2During an isothermal expansion, a confined ideal gas does $-150 \,J$ of work against its surroundings. This implies thatView Solution
- 3Two gases of equal mass are in thermal equilibrium. If ${P_a},\,{P_b}$ and ${V_a}$ and ${V_b}$ are their respective pressures and volumes, then which relation is trueView Solution
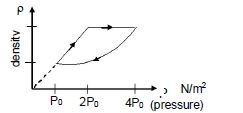
- 4An ideal gas undergoes cyclic process as shown in density pressure graph. During the process $AB$ the work done $|W_{AB}| = 70\,J$ . During the process $BC$, the gas absorbs $150\,J$ of heat. During the process $CA$ , gas undergoes expansion and does $210\,J$ of workView Solution
- 5Heat energy of $735\,J$ is given to a diatomic gas allowing the gas to expand at constant pressure. Each gas molecule rotates around an internal axis but do not oscillate. The increase in the internal energy of the gas will be $..........\,J$View Solution
- 6$V\, = \,K\,{\left( {\frac{P}{T}} \right)^{0.33}}$ where $k$ is constant. It is an,View Solution
- 7Work done by $0.1$ mole of a gas at ${27^o}C$ to double its volume at constant pressure is ....... $cal$ ($R = 2 \,cal\, mol^{-1}$ $^oC^{-1}$)View Solution
- 8A refrigerator works between $4^o C$ and $30^o C.$ It is required to remove $600$ calories of heat every second in order to keep the temperature of the refrigerated space constant. The power required is ....... $W$ (Take $1\, cal \,=\, 4.2\, Joules\,)$View Solution
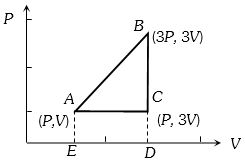
- 9An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution
- 10An ideal gas undergoes a polytropic given by equation $P V^n=$ constant. If molar heat capacity of gas during this process is arithmetic mean of its molar heat capacity at constant pressure and constant volume then value of $n$ is ..............View Solution
