Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
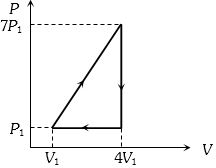
- 1View SolutionDuring an isothermal expansion of an ideal gas
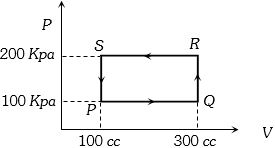
- 2A thermodynamic system is taken through the cycle $PQRSP$ process. The net work done by the system is ....... $J$View Solution
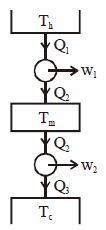
- 3Suppose that two heat engines are connected in series, such that the heat exhaust of the first engine is used as the heat input of the second engine as shown in figure. The efficiencies of the engines are $\eta_1$ and $\eta_2$, respectively. The net efficiency of the combination is given byView Solution
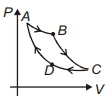
- 4Carnot cycle is plotted in $P-V$ graph. Which portion represents an isothermal expansion?View Solution
- 5Air is filled in a motor tube at ${27^o}C$ and at a pressure of $8$ atmospheres. The tube suddenly bursts, then temperature of air is $[{\rm{Given}}\,\,\gamma \,{\rm{of}}\,{\rm{air}} = \,1.5]$View Solution
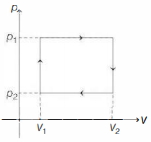
- 6$n$ moles of a van der Waals' gas obeying the equation of state $\left(p+\frac{n^2 a}{V^2}\right)(V-n b)=n R T$, where $a$ and $b$ are gas dependent constants, is made to undergo a cyclic process that is depicted by a rectangle in the $p-V$ diagram as shown below. What is the heat absorbed by the gas in one cycle?View Solution
- 7If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution
- 8A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases isView Solution
- 9During an experiment, an ideal gas is found to obey a condition $\frac{{{P^2}}}{\rho }$ = constant [$\rho =$ density of the gas]. The gas is initially at temperature $T,$ pressure $P$ and density $\rho$ . The gas expands such that density changes to $\rho/2$View Solution
- 10Which of the following graphs correctly represents the variation of $\beta = - (dV/dP)/V$ with $P$ for an ideal gas at constant temperatureView Solution