A gas is allowed to expand isothermally. The root mean square velocity of the molecules
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two non-reactive monoatomic ideal gases have their atomic masses in the ratio $2: 3$. The ratio of their partial pressures, when enclosed in a vessel kept at a constant temperature, is $4: 3$. The ratio of their densities is:View Solution
- 2The air density at Mount Everest is less than that at the sea level. It is found by mountaineers that for one trip lasting a few hours, the extra oxygen needed by them corresponds to $30,000\ cc$ at sea level (pressure $1$ atmosphere, temperature $27^oC).$ Assuming that the temperature around Mount Everest is $-73^oC$ and that the oxygen cylinder has capacity of $5.2\ litre,$ the pressure at which $O_2$ be filled (at site) in cylinder is ..... $atm$View Solution
- 3$\frac{1}{2} $ mole of helium gas is contained in a container at $S.T.P.$ The heat energy needed to double the pressure of the gas, keeping the volume constant (specific heat of the gas $ = 3\,J\,g{m^{ - 1}}\,{K^{ - 1}})$ is ...... $J$View Solution
- 4From the following $P-T$ graph what interference can be drawnView Solution
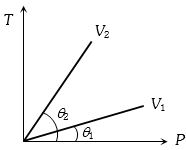
- 5At which of the following temperature would the molecules of a gas have twice the average kinetic energy they have at $20°C$View Solution
- 6When $2 \,\,gms$ of a gas are introduced into an evacuated flask kept at $25\,^0C$ the pressure is found to be one atmosphere. If $3 \,\,gms$ of another gas added to the same flask the pressure becomes $1.5$ atmospheres. The ratio of the molecular weights of these gases will beView Solution
- 7$Assertion :$ At a given temperature the specific heat of a gas at constant pressure. is always greater than its specific heat at constant volumeView Solution
$Reason :$ When a gas is heated at constant volume some extra heat is needed compared to that at constant pressure for doing work in expansion.
- 8Let $\gamma_1$ be the ratio of molar specific heat at constant pressure and molar specific heat at constant volume of a monoatomic gas and $\gamma_2$ be the similar ratio of diatomic gas. Considering the diatomic gas molecule as a rigid rotator, the ratio, $\frac{\gamma_1}{\gamma_2}$ isView Solution
- 9View SolutionWhich one the following graphs represents the behaviour of an ideal gas
- 10Three vessels of equal volume contain gases at the same temperature and pressure. The first vessel contains neon (monoatomic), the second contains chlorine (diatomic) and third contains uranium hexafloride (polyatomic). Arrange these on the basis of their root mean square speed $\left(v_{ ms }\right)$ and choose the correct answer from the options given below:View Solution
