Using equipartition of energy, the specific heat (in $J\, kg^{-1}\, K^{-1}$ ) of aluminium at room temperature can be estimated to be ( atomic weight of aluminium $= 27$)
JEE MAIN 2015, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The specific heat of a gas at constant volume is $21.2\, J/mole/°C.$ If the temperature is increased by $1°C$ keeping the volume constant, the change in its internal energy will be ...... $J$View Solution
- 2Pressure versus temperature graph of an ideal gas at constant volume $V$ of an ideal gas is shown by the straight line $A$. Now mass of the gas is doubled and the volume is halved, then the corresponding pressure versus temperature graph will be shown by the lineView Solution
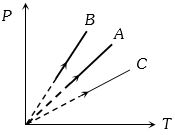
- 3View SolutionSelect the incorrect relation. (Where symbols have their usual meanings)
- 4At ...... $ ^o C$ temperature, the mean kinetic energy of ${O_2}$ will be the same for ${H_2}$ molecules at $- 73^o C$View Solution
- 5The number of gas molecules striking per second per square metre of the top surface of a table placed in a room at $20^{\circ} C$ and 1 atmospheric pressure is of the order of $\left(k_{B}=1.4 \times 10^{-23} \,JK ^{-1}\right.$ and the average mass of an air molecule is $5 \times 10^{-27} \,kg$ )View Solution
- 6For a certain process, pressure of diatomic gas varies according to the relation $P = aV^2$, where $a$ is constant. What is the molar heat capacity of the gas for this process?View Solution
- 7What will be the average value of energy along one degree of freedom for an ideal gas in thermal equilibrium at a temperature $T \,?\left( k _{ B }\right.$ is Boltzmann constant)View Solution
- 8$1\ mole$ of a gas with $\gamma$ = $7/5$ is mixed with $1\ mole$ of a gas with $\gamma$ = $5/3$, then the value of $\gamma$ for the resulting mixture isView Solution
- 9Five particles have speeds $1, 2, 3, 4, 5 \,\,m/s$. the average velocity of the particles is (in $m/s$)View Solution
- 10Nitrogen gas is at $300^{\circ} C$ temperature. The temperature (in $K$) at which the $rms$ speed of a $H _{2}$, molecule would be equal to the $rms$ speed of a nitrogen molecule, is........View Solution
(Molar mass of $N _{2}$ gas $28\, g$ )
