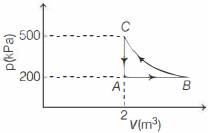
Six moles of an ideal gas perfomrs a cycle shown in figure. If the temperature are $T_A = 600 K, T_B = 800 K, T_C = 2200 K \,and\, T_D = 1200 K$, the work done per cycle is ..... $kJ$

Diffcult
(c) Processes $A$ to $B$ and $C$ to $D$ are parts of straight line graphs of the form $y = mx$
Also $P = \frac{{\mu R}}{V}T$ ($\mu = 6$)
==> $P \propto T.$ So volume remains constant for the graphs $AB$ and $CD$
So no work is done during processes for $A$ to $B$ and $C$ to $D$ i.e., $W_{AB} = W_{CD} = 0$ and $W_{BC} = P_2(V_C -V_B) = µR (TC -TB)$
$= 6R (2200 -800) = 6R \times 1400 J$
Also $ W_{DA} = P_1 (V_A -V_D) = µR(T_A -T_B)$
$= 6R (600 -1200)= -6R \times 600 J$
Hence work done in complete cycle
$W = W_{AB} + W_{BC} + W_{CD} + W_{DA}$
$= 0 + 6R \times 1400 + 0 -6R \times 600$
$= 6R \times 900 = 6 \times 8.3 \times 800 ≈ 40 kJ$
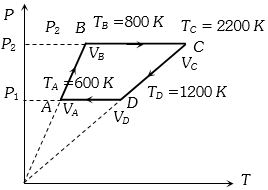
Also $P = \frac{{\mu R}}{V}T$ ($\mu = 6$)
==> $P \propto T.$ So volume remains constant for the graphs $AB$ and $CD$
So no work is done during processes for $A$ to $B$ and $C$ to $D$ i.e., $W_{AB} = W_{CD} = 0$ and $W_{BC} = P_2(V_C -V_B) = µR (TC -TB)$
$= 6R (2200 -800) = 6R \times 1400 J$
Also $ W_{DA} = P_1 (V_A -V_D) = µR(T_A -T_B)$
$= 6R (600 -1200)= -6R \times 600 J$
Hence work done in complete cycle
$W = W_{AB} + W_{BC} + W_{CD} + W_{DA}$
$= 0 + 6R \times 1400 + 0 -6R \times 600$
$= 6R \times 900 = 6 \times 8.3 \times 800 ≈ 40 kJ$

Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
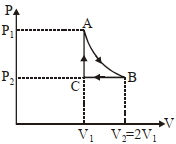
- 1$n$ mole a perfect gas undergoes a cyclic process $ABCA$ (see figure) consisting of the following processes.View Solution
$A \rightarrow B :$ Isothermal expansion at temperature $T$ so that the volume is doubled from $V _{1}$ to $V _{2}=2 V _{1}$ and pressure changes from $P _{1}$ to $P _{2}$
$B \rightarrow C :$ Isobaric compression at pressure $P _{2}$ to initial volume $V _{1}$
$C \rightarrow A$ : Isochoric change leading to change of pressure from $P _{2}$ to $P _{1}$
Total workdone in the complete cycle $ABCA$ is
- 2View SolutionWhen you make ice cubes, the entropy of water
- 3If $150 J$ of heat is added to a system and the work done by the system is $110 J$, then change in internal energy will be .......... $J$View Solution
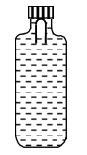
- 4A soft plastic bottle, filled with water of density $1 gm / cc$, carries an inverted glass test-tube with some air (ideal gas) trapped as shown in the figure. The test-tube has a mass of $5 gm$, and it is made of a thick glass of density $2.5 gm / cc$. Initially the bottle is sealed at atmospheric pressure $p_0=10^5 Pa$ so that the volume of the trapped air is $v_0=3.3 cc$. When the bottle is squeezed from outside at constant temperature, the pressure inside rises and the volume of the trapped air reduces. It is found that the test tube begins to sink at pressure $P_0+\Delta p$ without changing its orientation. At this pressure, the volume of the trapped air is $v_0-\Delta v$.View Solution
Let $\Delta v=X$ cc and $\Delta p=Y \times 10^3 Pa$.
($1$) The value of $X$ is
($2$) The value of $Y$ is
Give the answer or quetion ($1$) and ($2$)
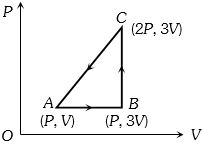
- 5An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution
- 6A diatomic gas $(\gamma=1.4)$ does $400 J$ of work when it is expanded isobarically. The heat given to the gas in the process is ............ $J$View Solution
- 7A Carnot freezer takes heat from water at $0\,^oC$ inside it and rejects it to the room at a temperature of $27\,^oC$. The latent heat of ice is $336 \times 10^3\, J\,kg^{-1}$. lf $5\, kg$ of water at $0\,^oC$ is converted into ice at $0\,^oC$ by the freezer, then the energy consumed by the freezer is close toView Solution
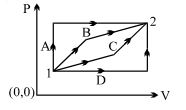
- 8An ideal gas is taken from state $1$ to state $2$ through optional path $A, B, C \& D$ as shown in $P-V$ diagram. Let $Q, W$ and $U$ represent the heat supplied, work done $\&$ internal energy of the gas respectively. ThenView Solution
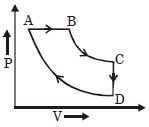
- 9A cyclic process $ABCD$ is shown in the given $P-V$ diagram. $P-T$ diagram that represents the same process isView Solution
- 10A constant amount of an ideal gas undergoes the cyclic process $A B C A$ in the $p-V$ graph shown below. The path $B C$ is an isothermal. The work done by the gas during one complete cycle, beginning and ending at $A$ is nearly .......... $\,kJ$View Solution