A sample of ideal gas $(\gamma = 1.4)$ is heated at constant pressure. If an $100\, J$ is supplied to gas, the work done by the gas is ...... $J$
Medium
At constant pressure
$\mathrm{W}=\mathrm{P} \Delta \mathrm{V}=\mathrm{n} \mathrm{R} \Delta \mathrm{T}$
But $Q=n C_{p} \Delta T=n \frac{\gamma R}{r-1}$
$\Rightarrow \quad W=Q\left(\frac{\gamma-1}{r}\right)=100\left(\frac{1.4-1}{1.4}\right)=28.57 \mathrm{J}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will beView Solution
- 2The amount of work done in an adiabatic expansion from temperature $T$ to ${T_1}$ isView Solution
- 3Agas expands such that its initial and final temperature are equal. Also, the process followed by the gas traces a straight line on the $P-V$ diagram :View Solution
- 4View SolutionWhich is incorrect
- 5A balloon filled with helium $\left(32^{\circ} C \right.$ and $1.7\; atm$.) bursts. Immediately afterwards the expansion of helium can be considered asView Solution
- 6One mole of ideal gas undergoes a linear process as shown in the figure below. Its temperature expressed as a function of volume $V$ isView Solution
- 7Choose the incorrect statement from the followingView Solution
$S1:$ The efficiency of a heat engine can be $1,$ but the coefficient of performance of a refrigerator can never be infinity.
$S2:$ The first law of thermodynamics is basically the principle of conservation of energy.
$S3:$ The second law of thermodynamics does not allow several phenomena consistent with the first law.
$S4:$ A process,whose only result is to transfer heat from a colder to a hotter object, is impossible.
- 8View SolutionIn thermodynamic processes which of the following statements is not true?
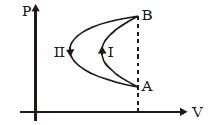
- 9In a cyclic process, a gas is taken from state $A$ to $B$ via path $-I$ as shown in the indicator diagram and taken back to state $A$ from state $B$ via path $-II$ . In the complete cycleView Solution
- 10A closed container is fully insulated from outside. One half of it is filled with an ideal gas $X$ separated by a plate Pfrom the other half $Y$ which contains a vacuum as shown in figure. When $P$ is removed, $X$ moves into $Y$. Which of the following statements is correct?View Solution