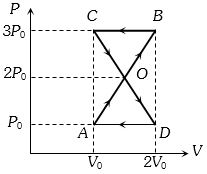
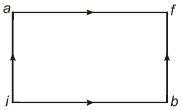
A thermodynamic system undergoes cyclic process $ABCDA $ as shown in figure. The work done by the system in the cycle is

AIPMT 2014, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
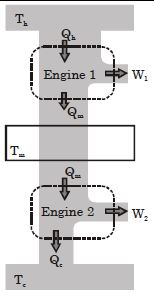
- 1Suppose that two heat engines are connected in series, such that the heat released by the first engine is used as the heat absorbed by the second engine, as shown in figure. The efficiencies of the engines are $\in_1$ and $\in_2$, respectively. The net efficiency of the combination is given by :View Solution
- 2View SolutionA gas is being compressed adiabatically. The specific heat of the gas during compression is
- 3A thermodynamic process in which temperature $T$ of the system remains constant though other variable $P$ and $V$ may change, is calledView Solution
- 4The gas law $\frac{{PV}}{T} = $ constant is true forView Solution
- 5$P_i, V_i$ and $P_f$, $V_f$ are initial and final pressures and volumes of a gas in a thermodynamic process respectively. If $PV^n =$ constant, then the amount of work done isView Solution
- 6The adiabatic elasticity of hydrogen gas $(\gamma = 1.4)$ at $NTP$ isView Solution
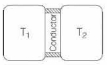
- 7A thin piece of thermal conductor of constant thermal conductivity insulated on the lateral sides connects two reservoirs which are maintained at temperatures $T_{1}$ and $T_{2}$ as shown in the figure alongside. Assuming that the system is in steady state, which of the following plots best represents the dependence of the rate of change of entropy on the ratio of $T_{1} / T_{2}$ ?View Solution
- 8A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)View Solution
- 9When a system is taken from state $i$ to state $f$ along the path $iaf$, it is found that $Q=50$ $cal$ and $W=20$ $cal$ Along the path $ibf\ Q = 36\ cal. \ W$ along the path $ibf$ is ....... $ cal$View Solution
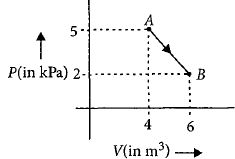
- 10One mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure.View Solution
The change in internal energy of the gas during the transition is ............$\;kJ$