The efficiency of carnot engine is $50\%$ and temperature of sink is $500\;K$. If temperature of source is kept constant and its efficiency raised to $60\%$, then the required temperature of the sink will be
AIPMT 2002, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Air is filled in a motor tube at ${27^o}C$ and at a pressure of $8$ atmospheres. The tube suddenly bursts, then temperature of air is $[{\rm{Given}}\,\,\gamma \,{\rm{of}}\,{\rm{air}} = \,1.5]$View Solution
- 2A Carnot engine whose low temperature reservoir is at $7\,°C$ has an efficiency of $50\%$. It is desired to increase the efficiency to $70\%$. By how many degrees should the temperature of the high temperature reservoir be increased ....... $K$View Solution
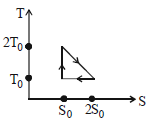
- 3View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
- 4A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases isView Solution
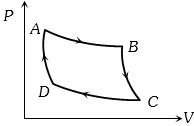
- 5Carnot cycle (reversible) of a gas represented by a Pressure-Volume curve is shown in the diagramConsider the following statementsView Solution
$I.$ Area $ABCD =$ Work done on the gas
$II.$ Area $ABCD =$ Net heat absorbed
$III.$ Change in the internal energy in cycle $= 0$
Which of these are correct
- 6Heat is supplied to a diatomic gas at constant pressure. The ratio of $\Delta Q\,:\,\Delta U\,:\,\Delta W$ isView Solution
- 7Given below are two statements:View Solution
Statement $I:$ If heat is added to a system, its temperature must increase.
Statement $II:$ If positive work is done by a system in a thermodynamic process, its volume must increase.
In the light of the above statements, choose the correct answer from the options given below
- 8The change in the entropy of a $1$ mole of an ideal gas which went through an isothermal process from an initial state $(P_1, V_1,T)$ to the final state $(P_2, V_2,T)$ is equal toView Solution
- 9$200\,g$ water is heated from $40\,^oC$ to $60\,^oC.$ Ignoring the slight expansion of water, the change in its internal energy is close to ...... $kJ$ (Given specific heat of water $=4184\,J/kgK$ )View Solution
- 10View SolutionWhich of the following statements is correct for any thermodynamic system
