If $150 J$ of heat is added to a system and the work done by the system is $110 J$, then change in internal energy will be .......... $J$
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$300 \,cal$. of heat is given to a heat engine and it rejects $225 \,cal$. of heat. If source temperature is $227^{\circ} C$, then the temperature of sink will be____${ }^{\circ} C$.View Solution
- 2View SolutionIn Carnot engine the work done by working substance is equivalent to
- 3An ideal gas has volume ${V_0}$ at ${27^o}C.$ It is heated at constant pressure so that its volume becomes $2{V_0}.$ The final temperature isView Solution
- 4View SolutionWhich relation is correct for isometric process
- 5View SolutionFirst law of thermnodynamics is given by
- 6The $P-V$ diagram shows seven curved paths (connected by vertical paths) that can be followed by a gas. Which two of them should be parts of a closed cycle if the net work done by the gas is to be at its maximum valueView Solution
- 7An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
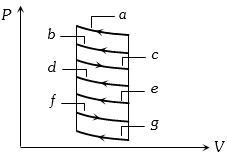
- 8One mole of an ideal gas undergoes two different cyclic processes I and II, as shown in the $P-V$ diagrams below. In cycle I, processes $a, b, c$ and $d$ are isobaric, isothermal, isobaric and isochoric, respectively. In cycle II, processes $a^{\prime}, b^{\prime}, c^{\prime}$ and $d^{\prime}$ are isothermal, isochoric, isobaric and isochoric, respectively. The total work done during cycle I is $W_I$ and that during cycle II is $W_{I I}$. The ratio $W_I / W_{I I}$ is . . . .View Solution
(image)
- 9Areversible adiabatic path on a $P-V$ diagram for an ideal gas passes through stateAwhere $P=0$.$7\times 10^5 \,\,N/ m^{-2}$ and $v = 0.0049 \,\,m^3$. The ratio of specific heat of the gas is $1.4$. The slope of path at $A$ is :View Solution
- 10An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution