Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
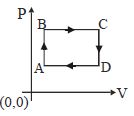
- 1The figure shows $P-V$ diagram of a thermodynamic cycle. Which corresponding curve is correct?View Solution
- 2The volume of an ideal gas $(\gamma=1.5)$ is changed adiabatically from $5$ litres to $4$ litres. The ratio of initial pressure to final pressure is:View Solution
- 3A cylinder of mass $1\,kg$ is given heat of $20000\, J$ at atmospheric pressure. If initially temperature of cylinder is $20\,^oC$, then work done by the cylinder will be .......$J$ (Given that Specific heat of cylinder $= 400 \,J\, kg^{-1}$, Coefficient of volume expansion $= 9 \times {10^{-5}}\,^o C^{-1}$, Atmospheric pressure $= 10^5 \,N/m^2$ and density of cylinder $9000\,kg/m^3$)View Solution
- 4A Carnot engine whose sink is at $300\, K$ has an efficiency of $40\%.$ By how much should the temperature of source be increased so as to increase its efficiency by $50\%$ of original efficiency ..... $K$View Solution
- 5View SolutionThe isothermal bulk modulus of a perfect gas at normal pressure is
- 6A reversible engine has an efficiency of $\frac{1}{4}$. If the temperature of the sink is reduced by $58^{\circ} {C}$, its efficiency becomes double. Calculate the temperature of the sink. (In $^{\circ} {C}$)View Solution
- 7The $P-V$ diagram of a certain process (carnot cycle) is as shown. The process is also represent asView Solution
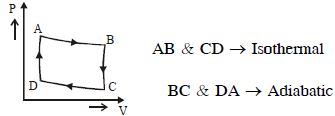
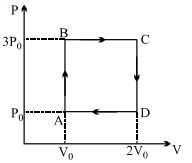
- 8A ideal monoatomic gas is carried around the cycle $ABCDA$ as shown in the fig. The efficiency of the gas cycle isView Solution
- 9A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
- 10A scientist says that the efficiency of his heat engine which operates at source temperature $127°C$ and sink temperature $27°C$ is $26\%$, thenView Solution
