A coil of resistance $100\Omega$ is connected across a battery of emf $6.0V.$ Assume that the heat developed in the coil is used to raise its temperature. If the heat capacity of the coil is $4.0J/K,$ how long will it take to raise the temperature of the coil by $15^\circ C$?
$\text{R}=100\Omega,$
$\text{E}=6\text{v}$
Heat capacity of the coil $=4\text{J/k}$
$\Delta\text{T}=15^\circ$
Heat liberate $\Rightarrow\frac{\text{E}^2}{\text{Rt}}=4\text{J/K}\times15$
$\Rightarrow\frac{6\times6}{100}\times\text{t}=60$
$\Rightarrow\text{t}=166.67\text{sec}=2.8\text{min}$
$\text{E}=6\text{v}$
Heat capacity of the coil $=4\text{J/k}$
$\Delta\text{T}=15^\circ$
Heat liberate $\Rightarrow\frac{\text{E}^2}{\text{Rt}}=4\text{J/K}\times15$
$\Rightarrow\frac{6\times6}{100}\times\text{t}=60$
$\Rightarrow\text{t}=166.67\text{sec}=2.8\text{min}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Given $n$ resistors each of resistance $$R, how will you combine them to get the $(i)$ maximum $(ii)$ minimum effective resistance? What is the ratio of the maximum to minimum resistance?View Solution
- 2View SolutionIn the given circuit, with steady current, calculate the potential drop across the capacitor and the charge stored in it.
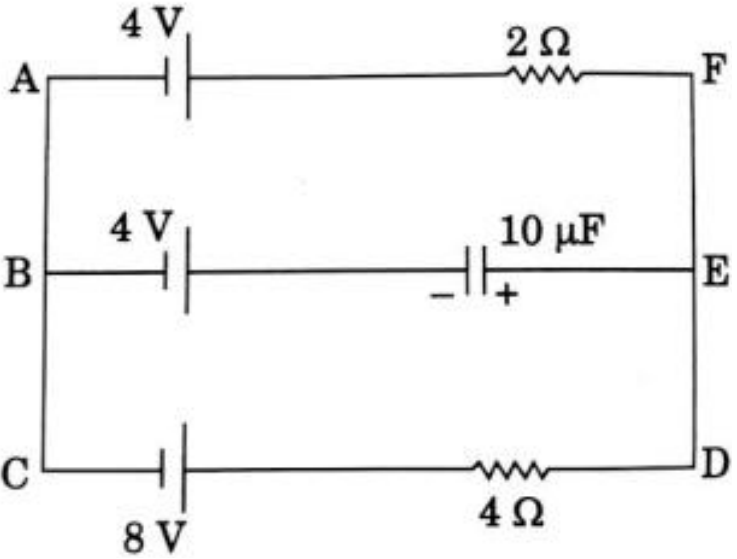
- 3View SolutionIn the given circuit, with steady current, calculate the potential drop across the capacitor in terms of V.
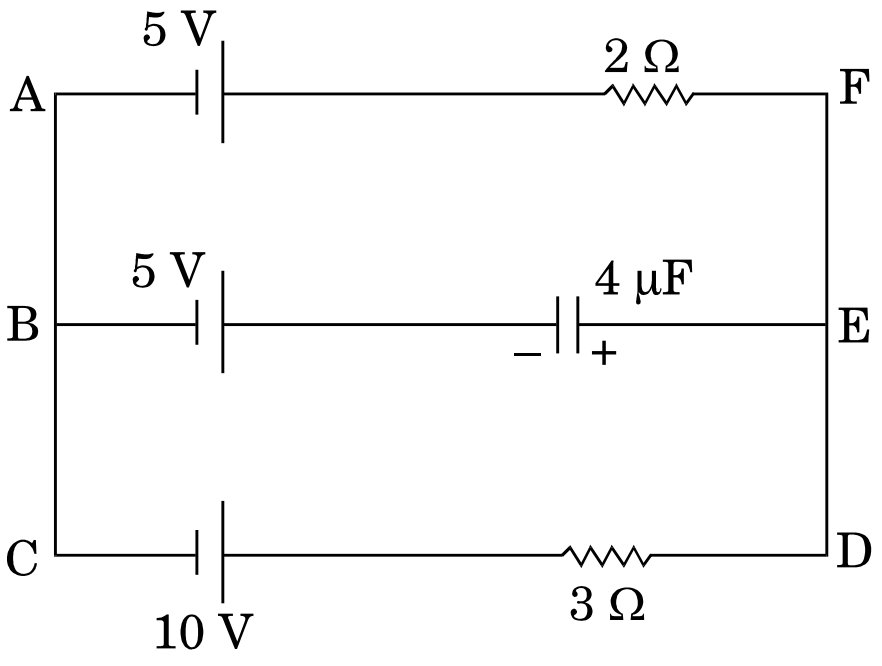
- 4A resistance of $R \Omega$ draws current from a potentiometer as shown in the figure. The potentiometer has a total resistance $R_o\Omega$ . A voltage $V$ is supplied to the potentiometer. Derive an expression for the voltage across $R$ when the sliding contact is in the middle of the potentiometer.View Solution
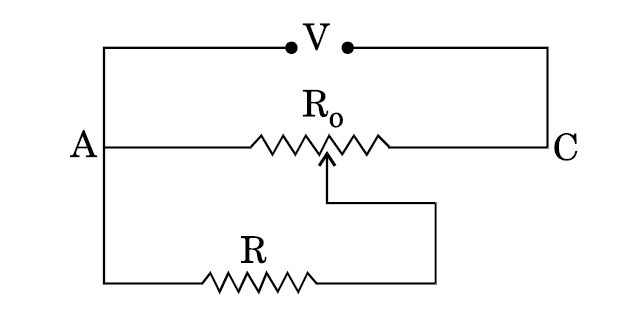
- 5View SolutionA (i) series (ii) parallel combination of two given resistors is connected, one by one, across a cell. In which case will the terminal potential difference, across the cell have a higher value?
- 6Using Kirchhoff’s rules, calculate the current through the $40\Omega$ and $20\Omega$ resistors in the following circuit:View Solution
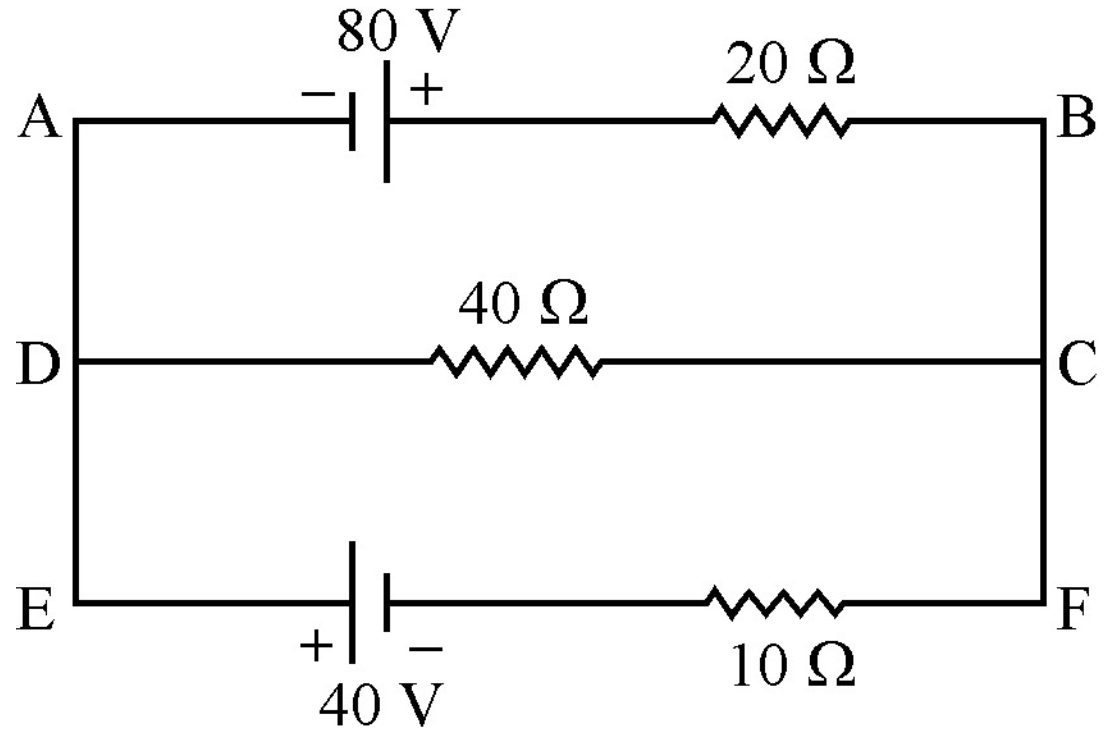
- 7View SolutionIn the given circuit, with steady current, calculate the potential difference across the capacitor and the charge stored in it.
- 8The current through a wire depends on time as $\text{i}=\text{i}_0+\alpha\text{t},$View Solution
Where $\text{i}_0=10\text{A}$ and $\alpha=4\text{A/ s}.$ Find the charge that crosses through a section of the wire in 10 seconds. - 9A plate of area $10\ cm^2$ is to be electroplated with copper $($density $9000\ kg/m^3)$ to a thickness of $10$ micrometres on both sides, using a cell of $12V$. Calculate the energy spent by the cell in the process of deposition. If this energy is used to heat $100g$ of water, calculate the rise in the temperature of the water. $\ce{ECE}$ of copper $= 3 \times 10^{-7}kg C^1 $ and specific heat capacity of water $= 4200Jkg^1.$View Solution
- 10View SolutionWhat is the advantage of using thick metallic strips to join wires in a potentiometer?
