A vessel is partitioned in two equal halves by a fixed diathermic separator. Two different ideal gases are filled in left $(L)$ and right $(R)$ halves. The rms speed of the molecules in $L$ part is equal to the mean speed of molecules in the $R$ part. Then the ratio of the mass of a molecule in $L$ part to that of a molecule in $R$ part is


Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
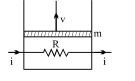
- 1A resistance coil connected to an external battery is placed inside an adiabatic cylinder fitted with a frictionless pistn and containing an ideal gas. A current $i$ flows through the coil which has a resistance $R$. At what speed must the piston move upward in order that the temperature of the gas remains uchanged? Neglect atmospheric pressure.View Solution
- 2An object is placed in a medium of refractive index $3$. An electromagnetic wave of intensity $6 \times 10^8 \mathrm{~W} / \mathrm{m}^2$ falls normally on the object and it is absorbed completely. The radiation pressure on the object would be (speed of light in free space $=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ ):View Solution
- 3Statement$-1 :$ Internal energy of gas $U = nC_VT$ is due to random motion of gas molecules.View Solution
Statement$-2 :$ A container is moving with speed $v$. It is suddenly stopped by a force, temperature of gas increases. - 4If $10^{22}$ gas molecules each of mass $10^{-26}\, kg$ collide with a surface (perpendicular to it)elastically per second over an area $1\, m^2$ with a speed $10^4\,m/s$, the pressure exerted by the gas molecules will be of the order ofView Solution
- 5The temperature of a gas is $-50^{\circ}\,C$. To what temperature the gas should be heated so that the rms speed is increased by $3$ times?View Solution
- 6View SolutionMean kinetic energy (or average energy) per gm molecule of a monoatomic gas is given by
- 7Two containers $C_{1}$ and $C_{2}$ of volumes $V$ and $4 \,V$ respectively, hold the same ideal gas and are connected by a thin horizontal tube of negligible volume with a valve which is initially closed. The initial pressures of the gas in $C_{1}$ and $C_{2}$ are $p$ and $5 p$, respectively. Heat baths are employed to maintain the temperatures in the containers at $300 \,K$ and $400 \,K$, respectively. The valve is now opened. Select the correct statement.View Solution
- 8If the root mean square velocity of hydrogen molecule at a given temperature and pressure is $2 \mathrm{~km} / \mathrm{s}$, the root mean square velocity of oxygen at the same condition in $\mathrm{km} / \mathrm{s}$ is :View Solution
- 9A cylinder with fixed capacity of $67.2\, lit$ contains helium gas at $STP$. The amount of heat needed to raise the temperature of the gas by $20\,^oC$ is ..... $J$ [Given that $R = 8.31\, J\, mol^{-1}\, K^{-1}$]View Solution
- 10$S.I.$ unit of universal gas constant isView Solution
